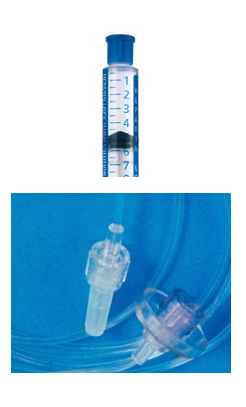
 The small solid cap found on the end of syringes, all type of I.V. administration sets, and add-on devices serves a tremendous purpose – keeping the sterile part from being contaminated. I encounter a lot of misunderstanding about these caps and how they should be used. For most administration sets, extension sets, add-on filters, and prefilled flush syringes, the sterile components include the fluid pathway and the ends of the device that is covered by that tip cap. Removal of that tip cap should only happen immediately before it is to be used. Some extension sets and prefilled syringes are completely sterile and can be added to a sterile field. Always read the label to make sure the entire device is sterile before dropping it onto a sterile field. Removing a tip cap prematurely creates an unnecessary hazard. The set or syringe can be dropped onto the bed, becoming contaminated. Or the nurse holds it while doing other things such as disconnecting the I.V. set from the catheter, stopping the infusion pump, scrubbing the connection surface, etc. This dramatically increases the risk of contaminating the device being held as it easily comes into contact with linens or clothing or the patient’s arm. Another dangerous practice is the cover on the port of a plastic IV fluid container. It takes some force to pull these covers off. Don’t hold the exposed spike end of the IV set in your hand when removing this cover. Your hand movement when the cover breaks loos can easily cause the spike to touch something and be contaminated. Remove the cover from the new bag first, then remove the spike from the empty bag or the cap from spike end. The safest method is the best method – do all other steps FIRST. Remove the tip cap immediately before connecting the set or syringe so that the connection is made with a sterile device. Tip caps are definitely a single use item. Once they have been removed from the original device it is covering, it goes in the trash. They cannot be re-applied to a set or syringe. They cannot be used for other purposes such as placing the tip cap from the prefilled flush syringe on the male luer of the intermittent I.V. administration set. There is no label on the device indicating that the tip cap can be used for other purposes, so simply throw it away immediately. Cover the male luer end of a syringe or set ONLY with a new sterile tip cap. Misuse of tip caps and premature removal is one avenue to introduce organisms through the lumen of the catheter. Up to 45% of catheter hubs are contaminated with regular use, leading to intraluminal contamination and catheter related bloodstream infection. Vascular catheter infections of all kinds – bloodstream, local exit site, suppurative thrombophlebitis - causes significant losses for your employer. If this infection occurs while the patient is hospitalized, there is NO money paid to the hospital to treat the infection. The hospital is prohibited from billing this treatment to the patient. And hospitals that have high rates of central line associated bloodstream infection are penalized by having 1% to 2% of their annual Medicare payment take away. Do not be the cause of your patient’s infection. Do not cause your employer to loose money. Do not remove the tip cap prematurely. Do not contaminate the sterile component before using it. Preventing infection is in your hands! |
Author: Lynn HadawayLynn Hadaway is an international thought leader in infusion therapy and vascular access, having been in this practice for more than 40 years. Her experience comes from hospital-based infusion teams, device manufacturers, and continuing education services. Her journal and textbook publications are extensive. She also maintains board certification in infusion nursing (CRNI) and nursing professional development (RN-BC). Categories
Archives
May 2019
|
Copyright © 2020 Lynn Hadaway Associates, Inc. | P.O. Box 10, Milner, GA 30257 | 770-358-7861


 RSS Feed
RSS Feed